Psilocybin Treatment of PTSD
Investigating the safety and tolerability of single-dose psilocybin for post-traumatic stress disorder: A nonrandomized open-label clinical trial. McGowan et al. (2025). Journal of Psychopharmacology, 00 (0), 1-10. DOI: 10.1177/02698811251362390
Abstract
Background: Post-traumatic stress disorder (PTSD) is a debilitating condition for which there are few efficacious treatments. Psilocybin is being studied for use in treatment-resistant depression but has not yet been investigated in PTSD.
Aims: The trial’s primary outcome was to investigate the safety and tolerability of single-dose psilocybin in participants with PTSD.
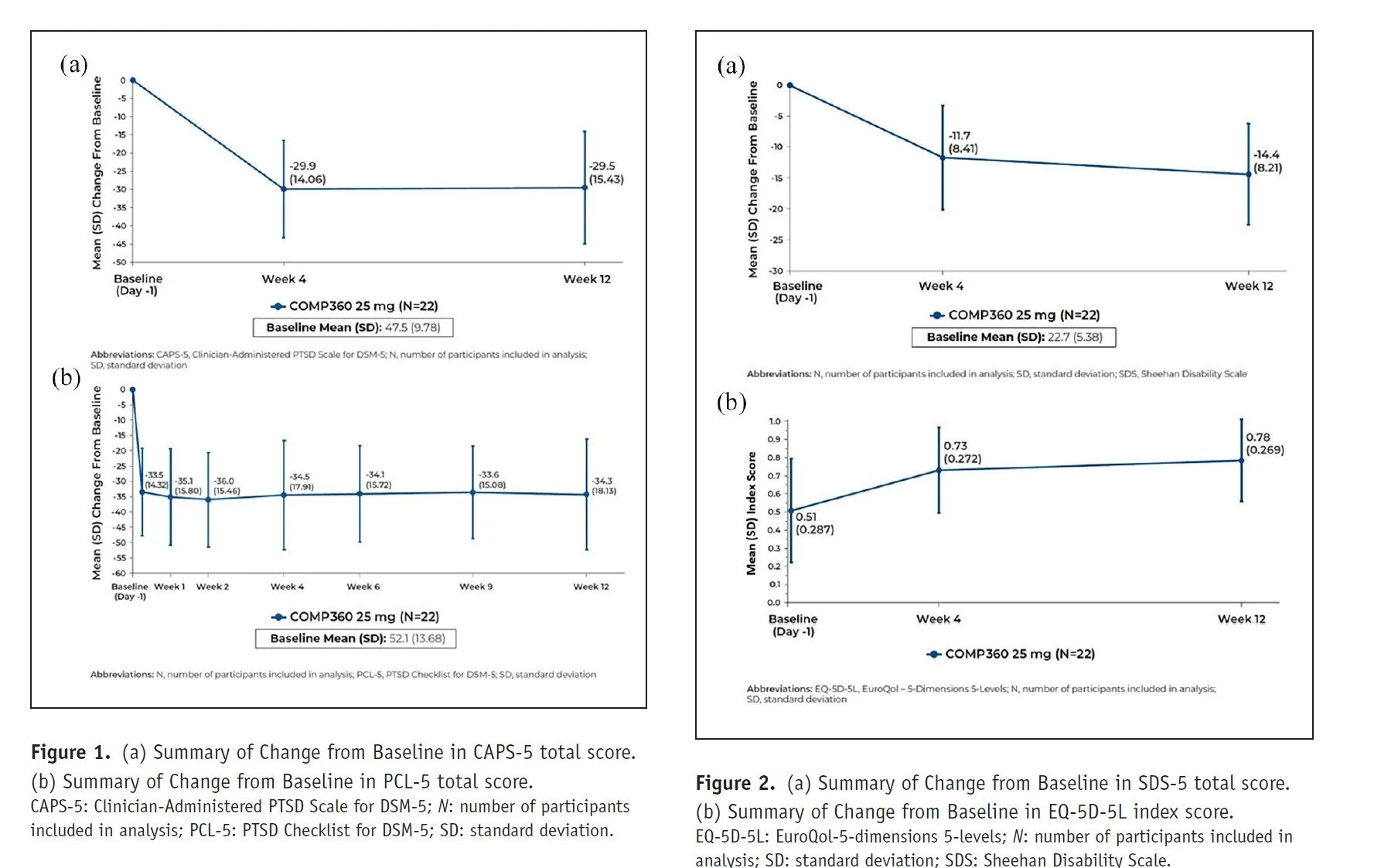
Methods: This was a Phase 2, nonrandomized, open-label, multicenter trial. 22 participants enrolled (63.6% female; mean (SD) age, 39.0 (7.91) years). Secondary outcomes were changes in PTSD symptoms (Clinician-Administered PTSD Scale for DSM-5 (CAPS-5); PTSD Checklist for DSM-5 (PCL-5)), functional impairment (Sheehan Disability Scale; SDS) and quality of life (EQ-5D-5L index score).
Results: Pre-post comparisons indicated a clinically meaningful change from Baseline in mean CAPS-5 total score at Week 4 (−29.9 (14.06)) and Week 12 (−29.5 (15.43)), which was associated with the intensity of psychedelic experience on Day 1. PCL-5 scores showed symptom reduction was rapid and sustained until Week 12. SDS total score and EQ-5D-5L index score showed similar improvements. There were no treatment-emergent adverse events (TEAEs) leading to study withdrawal.
Conclusions: Psilocybin at a dose of 25 mg, administered with psychological support, may be safe, well-tolerated and associated with symptomatic improvement in adults with PTSD.